 Include a table showing the name of the dye, total number of π-electrons, maximum wavelength, and box lengthĥ. Calculate the effective box length, a, for the dyesĤ. Plot the UV-Vis absorption spectra yourself (Do NOT just include a screen shot) and label the maximum wavelength for each dyeģ. Do not do the “theoretical calculations” sectionĢ. Complete the analysis questions found in the text book under experiment #34 a. In addition, these dyes slowly degrade in the presence of light, so attempt to keep the solutions stored in the dark when not in use.ġ. Be advised and avoid getting the solutions on your skin. *Safety note: The dyes being used in this experiment are toxic. Dispose of used methanol/dye solutions in the appropriate waste container Determine maximum wavelength for each dyeĦ. Record spectra against a solvent reference, scanning from 400-800 nmĥ. Please check your calculations in order to minimize the amount of solventģ. Recall Beer’s A = εcb & reference the extinction coefficientĢ. Prepare dilute solutions of select dyes such that the final concentration will give anĪbsorbance between 0.5 and 1.0 for a 1cm path lengthġ. Prepare stock solutions of the dyes with methanol as the solvent (approx. In this experiment, we shall assume that the wavelength maximum (the wavelength at which the dyes absorb most strongly) is the wavelength to use in the calculations.ġ. Although sharp lines are observed for isolated atoms, broad absorption bands are observed for substances in liquid phases (due to accompanying vibrational and rotational transitions). 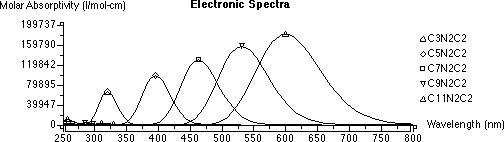 
*Note on wavelengths: If only changes in electronic energy accompany absorption of light, a very sharp maximum in absorption should be observed at the characteristic wavelength. This crude model of electrons moving along a chain of carbon atoms can be successfully modeled with the Particle-in-a-Box quantum mechanical model. By changing the size of the conjugated system, the effective “box” length over which the electrons can move is changed. These polyenes have a structure with alternating π-electron character (i.e., double bonds) in which the electrons are delocalized over the entire conjugated system. The bands for polyene dyes arise from electronic transitions involving the π- electrons along the chain of the molecule and the associated wavelength of these bands depends on the spacing of the electronic energy levels.Ĭonjugated polyenes, such as β-carotene, are ubiquitous pigments in nature and generally absorb light in the visible portion of the electromagnetic spectrum. In this experiment Ultraviolet-Visible spectroscopy is used to explore the electronic structure of several conjugated polyene dyes, and the Particle-in-a-Box model is used to extract structural information. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
